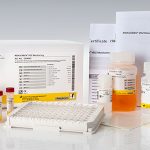
Intended use:
The RIDA®QUICK Rota/Adeno/Noro Combi is a single step immunochromatographic lateral flow assay for the differential qualitative detection of rotavirus, adenovirus and norovirus of genogroups I and II in human stool specimen.
General information:
Acute viral gastroenteritis is mainly caused by norovirus, rotavirus, and adenovirus. Infections with these viruses may appear all over the year. Whereas norovirus and rotavirus have a seasonal pattern with increasing infections during winter months, adenovirus infections are slightly more common in the summer months.
Several studies report increasing numbers of viral co-infections in patients with diarrhea who might suffer from a more severe disease. Moreover, symptoms of patients with acute diarrhea are not sufficiently specific to allow for a distinct diagnosis. Thus, rapid detection of the disease causing pathogen supports infection control and patient care.
RIDA®QUICK Rota/Adeno/Noro Combi combines detction of the most important viral pathogens in one test:
- Rapid detection of co-infections and outbreaks in less than 20 min
- Distinction between Norovirus genogroup I und genogroup II
- Simple workflow with ready for use materials
- Quality control material available
More information:
- Kaplon et al., 2019. Diagnostic Performance of Triplex Norovirus/Rotavirus/Adenovirus ICG tests.
- Kaplon et al., 2021. Diagnostic Accuracy of Four Commercial Triplex Immunochomatographic Tests for Rapid Detection of Rotavirus, Adenovirus, and Norovirus in Human Stool Samples.
Accessories:
| Art. No. | N1903 |
|---|---|
| Test format | Rapid test cassettes consisting of 20 tests |
| Sample preparation | Ready for use sample dilution tubes |
| Incubation time | 15 minutes test run |
Dear customers,
we have started to provide the documents for our products in an electronic format. These are the Instructions for Use (IFU), the Safety Data Sheets (SDS) and the Certificate of Analysis (CoA). For batches placed on the market after 01 January 2023, you can find our documents on the eIFU portal eifu.r-biopharm.com/IVD.